Overview
Hypothalamic hamartomas (HH) are lesions that arise in the ventral hypothalamic region while the brain is forming in utero.
The syndrome is commonly associated with a spectrum of clinical, endocrinologic, and psychosocial comorbidities, including refractory epilepsy, precocious puberty, and rage behaviors. Additionally, HH syndrome may include the evolution of an epileptic encephalopathy with developmental regression, psychiatric and behavioral comorbidities. Neuropsychological, sleep and endocrine disorders are also common.
With regards to epilepsy, for most, the initial seizures are gelastic seizures. Gelastic seizures usually begin early in life, often at less than 1 year of age, and are difficult to recognize. Gelastic seizures are the most specific symptom associated with HH and often appear as laughter (often mirthless) or giggles that an individual cannot control and happen without any obvious trigger. In the very young, they are often mistaken for colic, acid reflux or Irritable Bowel Syndrome (IBS). They can occur daily, and for some there may be hundreds per day. The epilepsy symptoms can progressively change, with the appearance of other seizure types such as infantile spasms, atypical absences, focal, generalized tonic-clonic seizures and drop- attacks. It is often not until these other seizure types manifest, that an HH is discovered. For many individuals, the process of getting a correct diagnosis can take months or years. Achieving an early and accurate diagnosis is critical and an appropriate surgical intervention is often recommended earlier in the treatment plan rather than a “wait and see” approach.
Incidence and prevalence
The prevalence of epilepsy in HH is estimated to be 1 in 200,000 individuals, with a slightly higher number of males versus females.
This estimate, which may be low since HH is hard to detect, suggests that there are about 30,000 in the world living with HH. Approximately 5% of all HH cases are associated with Pallister-Hall syndrome. There are likely to be people with epilepsy in whom the syndrome remains undiagnosed or misdiagnosed.

Etiology
Etiology of a hypothalamic hamartoma is not yet fully understood, but it is suspected that genetic factors contribute.
One large study has reported a sonic hedgehog gene abnormality (e.g. GLI3, PRKACA) in about one-third (~ 37%) of sporadic, non-syndromic cases. In most syndromic cases (with Pallister-Hall Syndrome), where molecular testing is performed, a genetic abnormality in the GLI3 gene is found to be the cause. There is emerging evidence that cilia gene (e.g.DYNC2H1) abnormalities can also cause HH in a significant minority of cases and research into cilia genes is on-going. This may mean that HH is ultimately reconceptualized as a ciliopathy. Genetic counseling is recommended for all Pallister-Hall cases, where the GLI3 abnormality can be transmitted from a parent to their child. It is not currently recommended for non-syndromic cases, however that may change depending on further research into the cilia gene abnormalities, as at least some of these can be transmitted. In both groups (non-syndromic or syndromic) of individuals with HH, determining the underlying genetic cause may have implications for future therapeutic approaches.
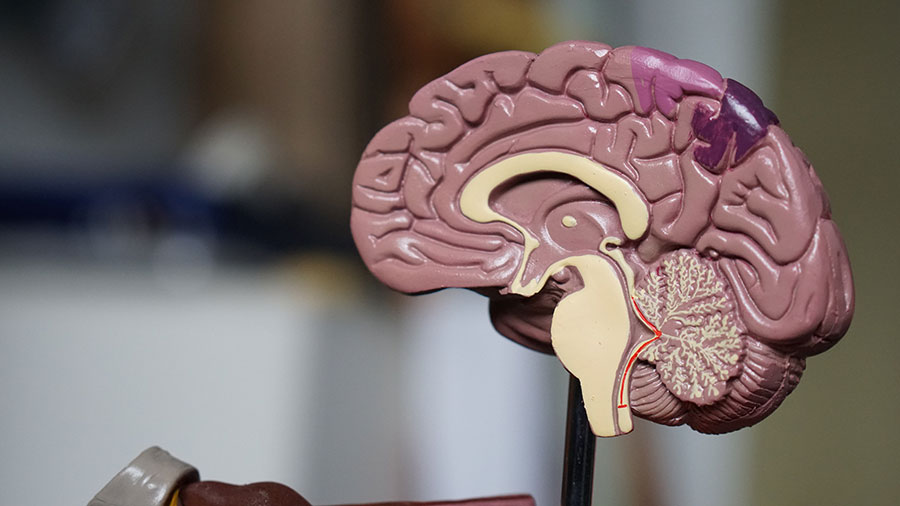
HH Lesion Types
Several attempts have been made to classify HH to better understand the characteristics and the impact the HH may have on the surrounding critical structures in that area of the brain.
What type of HH you have will be a factor when deciding on a treatment plan.
To properly understand hypothalamic hamartomas, a distinction must first be made between the 2 general subtypes or classifications. They are: 1) intrahypothalamic (or sessile) and 2) parahypothalamic (or pedunculated).
The intrahypothalamic (or sessile) HH has a base of attachment within the third ventricle and can be either partial or complete. These lesions vary significantly in size but typically extend into the third ventricle itself and distort the surrounding anatomical structures, most importantly, the fornix or mammilary body. These structures are critical in the process of memory and behavior and therefore it is common for short-term memory and behavior disturbances to be evident. Sessile HH’s are most often associated with neurological issues and an initial presentation of gelastic seizures with a progression to other seizure types. Just under half of individuals with sessile HH’s will develop precocious puberty at some point in their lifetime. It is not unusual for children with this type of HH to experience a progressive decline in behavior, cognitive abilities, and social skills.
The parahypothalamic (or pedunculated) HH is more often associated with precocious puberty and not usually associated with epilepsy or neurodevelopmental problems. Treatment for these types of lesions is usually aimed at controlling the effects of the sexual development (precocious puberty) and premature bone development. Most cases can be easily classified into one category or the other, but some combinations of the two do exist.
For a deeper dive into the description of HH lesions, go to A Closer Look for an excellent video and an expanded description of the Delalande and Fohen classification of hypothalamic hamartomas.
Diagnosis of HH
Diagnostic evaluation of HH remains challenging due to the depth of the lesion, which often results in a scalp EEG reading normal without any evidence of interictal abnormality.
Even a regular surface EEG during a well-known seizure type can be difficult to localize, or may be misleading, because by the time the seizure activity leaves the HH and makes its way to the brain surface, the EEG cannot “see” where it came from initially. The hamartoma can also be difficult to identify on imaging due to its small size and location. This is so even with experienced neuroradiologists, who may be more accustomed to searching for anatomical lesions in the cortex, the more typical location of seizure onset, rather than subcortical regions. The recommendation for locating and diagnosing a hypothalamic hamartoma is a high-resolution 3Tesla brain MRI with epilepsy specific protocol including: thin cut 3D T1 (≤ 1mm3 voxel) weighted sequences and T2 weighted and FLAIR sequences (minimum two planes, 3D better). MRI technicians often use the following protocol sequence parameters:
- 3D T1W SPGR, axial 0.9mm isotropic voxels
- Sag T1W – min TE; 3mm slice, 0.5mm gap; FOV 20cm
- Sag T2W(FSE) – 2mm slice no gap; FOV 20cm
- Cor T2W(FSE) – 2mm slice no gap; FOV 16cm
- Cor T1W – 3D SPGR; 2mm slice; FOV 24cm – recon for axial
- Axial T2W(FSE) – routine brain
The interpretation of this imaging often requires specific expertise, which is usually more readily available at skilled epilepsy centers.
Download the approved HH MRI Protocol
Age of seizure onset
Age of onset is typically less than a year for gelastic seizures, and between two to seven years of age for additional focal seizure types, both of which are often refractory to medical treatment.

Presentation of Seizure Types
Gelastic seizures are almost always the first seizure manifestation of HH.
Most individuals do not feel happy when experiencing a laughing seizure and in fact, may feel anxiety and panic when they are forced to laugh at inappropriate times. Other common features of a gelastic seizure are:
- The person may look startled or even have a look of panic or fear.
- There can also be an unpleasant feeling in the stomach (like butterflies), a tickling in the chest or headache. Lip smacking or frequent swallowing may be seen.
- The eyes may seem vacant and move up and to one side.
- There often is a slight smile that seems a bit forced and laughter or grunting that seems unusual or not appropriate at that time.
- May be triggered by loud noises or sudden actions.
- Often occur as the individual is falling off to sleep.
These seizures can occur many times a day. In some cases, over 100 a day. The seizure may present asymmetrically, with the stronger grimacing appearing on the side of the face contralateral to the HH attachment within the third ventricle. Sometimes crying (dacrystic) seizures may be seen.
For additional information regarding the presentation of gelastic seizures go to What is a Gelastic Seizure? page.
How seizure types change over time
Seizures often evolve to include focal seizures with impaired awareness and these may resemble those observed in temporal lobe epilepsy. Generalized seizures may also occur including atypical absences, tonic, atonic and generalized tonic-clonic seizures. Infantile spasms are occasionally seen.
Treatment
Anti-seizure medications (ASM), although necessary to reduce the risks of prolonged seizures, are usually not efficacious and overtreatment should be avoided.
Currently, there are several surgical options for HH; disconnection or ablation is the surgical aim. Which surgical approach to use is based upon location, size, and attachment of the hamartoma. Approaches include Gamma Knife radiosurgery, stereotactic radiofrequency thermocoagulation, MRI-guided laser therapy, endoscopic resection and transcallosal resection. MRI-guided focused ultrasound thermoablation is currently in clinical trials for HH. Appropriate surgical intervention can achieve total or partial seizure control but may not reverse encephalopathy.
Comorbidities
HH syndrome is associated with a varying comorbidity profile that includes neurodevelopmental, behavioral, endocrine and psychiatric dysfunction.
Precocious puberty presents in around a third of cases – and is often the clue that affects a timely diagnosis. Psychiatric comorbidities exist in over 50% of individuals. Rage attacks, as well as less severe aggressive behaviors and attention problems, are common. Cognitive impairments are also common, and these appear to be progressive in half of cases. In view of the apparent relationship between the epilepsy onset and neurocognitive difficulty, the syndrome is considered an epileptic encephalopathy, with increased seizure burden contributing to worse cognitive outcomes.
For additional information regarding other symptoms and comorbidities associated with the HH Syndrome go to The Symptoms page.
